Superiority
Heparinase I cleaves heparin and heparan sulfate (relative activity about 3:1) at the linkages between hexosamines and O-sulfated iduronic acids, yiel…
Details
Heparinase I cleaves heparin and heparan sulfate (relative activity about 3:1) at the linkages between hexosamines and O-sulfated iduronic acids, yielding mainly disaccharides. The enzyme also cleaves the antithrombin III binding pentasaccharide domain in the heparin molecule.
Recombinant Heparinase I from Flavobacterium heparinum; Host Cell: E. Coli.
Property
l pH stability
6.5-7.5
l Optimum pH
7.0
l Thermal stability
≤35℃(pH7.0, 20min)
l Optimum temperature
35℃
l Molecular weight
42,508
l Isoelectric point
9.4
Specification
l Specific activity >100 IU/mg.
Enzyme activity definition
One Heparinase I international unit (IU) is defined as the amount of enzyme that will liberate 1.0 μmole unsaturated oligosaccharides from porcine intestinal mucosal heparin per minute at 30℃ and pH 7.0.




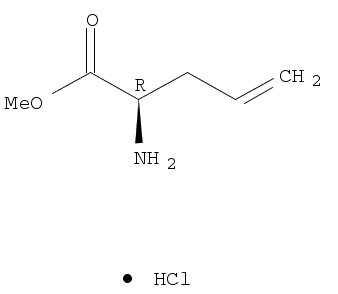
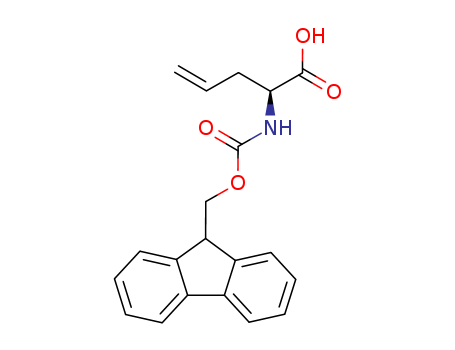
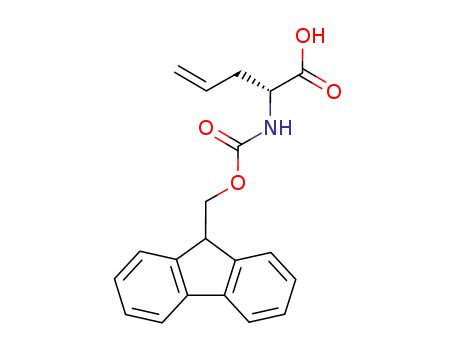
![4-Pentenoic acid, 2-[[(1,1-dimethylethoxy)carbonyl]amino]-](http://file1.lookchem.com/300w/synthetic/2022-02-05-02/dead4baa-ec50-4a61-9af9-08ebf07aafc8.png)